Kaunain Sheriff Mo
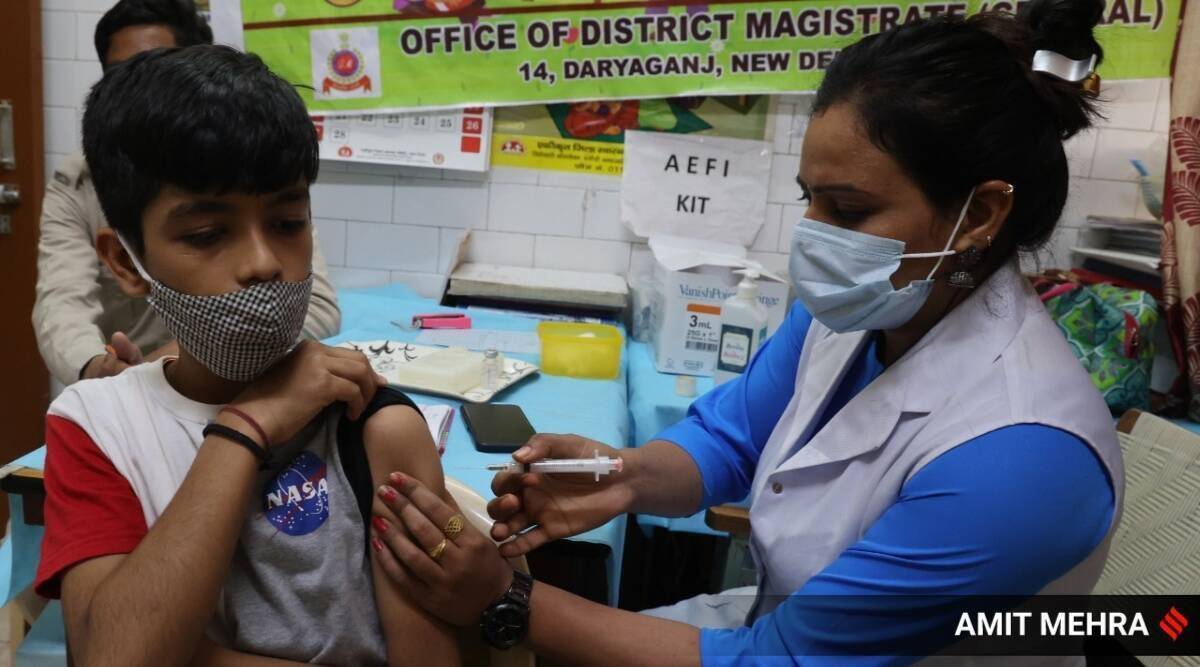
Health Ministry sources said the subject matter expert panel on COVID-19 vaccines on Thursday recommended India’s drug regulator to grant emergency use rights to Hyderabad-based organic E-Corbevax for the age group of 5-12 years. Top sources told The Indian Express that the recommendations of the subject expert committee for formal approval of the vaccine will be taken up by India’s drug regulator in the next few days.
With Thursday’s approval, the biological E-Corbevax will become the first COVID-19 vaccine candidate that can be administered to children over the age of 5, as the government expert body on COVID-19 recommends COVID-19 vaccination for this age group. Makes the final decision to start.
Corbevax vaccine is India’s first indigenously developed protein sub-unit vaccine against COVID-19. Currently, India is administering COVID-19 vaccines only to people in the age group of 12-18 years. Significantly, India is giving only Corbevax to the age group of 12-14 years. 2,53,87,677 have been given the first dose, and 12,47,298 have been fully vaccinated.
The Corbevax vaccine is built on the traditional subunit vaccine platform: instead of using the whole virus, the platform triggers an immune response using fragments of it, like the spike protein. The subunit vaccine contains the harmless S-protein – and once the human immune system recognizes the S protein, it produces antibodies in the form of white blood cells, which fight infection.
Bio e-Corbevax contains an antigen developed by the Texas Children’s Hospital Center for Vaccine Development and licensed from the Integrated Commercialization Team of BCM Ventures, Baylor College of Medicine. The Center has already made an advance payment of Rs 1500 crore to reserve 30 crore doses of Corbevax.
Meanwhile, most of the samples that were genome-sequenced in the first fortnight of April in Delhi showed the Omicron subtype BA.2.12 and this could be one of the reasons behind the recent rise in the number of COVID-19 cases in the city. . Sources said this on Thursday.
However, a source in the Indian SARS-CoV-2 Genomics Association (INSACOG) claimed that BA.2.12.1, derived from the variant of Omicron, was also detected in some samples from Delhi, which is being attributed to the recent increase in cases in the US. Is.